Definition of Molecules – Introduction to fundamental concepts of Biological Science including the organization and common characteristics of living matters, cell structures and functions, food production by photosynthesis, harvesting energy, mechanism of cells reproduction, genetics, evolutions, and Human Biology. Introduction to general chemistry including basic concepts about matter, atomic structure, chemical bonds, gases, liquid, and solids, solutions, chemical reactions, acid, bases, and salt;
organic and biochemistry including hydrocarbons and their derivatives, carbohydrates, lipids, proteins, enzymes, vitamins, and minerals, nucleic acids; principles of physics and applications to nursing including gravity and mechanics, pressure, heat and electricity; nuclear chemistry and nuclear physics, effects of radiation on human beings, and protection and disposal. The aim of the course is to acquire knowledge and skills in general biological science, general chemistry and physics.
Definition of Molecules
A molecule is the smallest particle in a chemical element or compound that has the chemical properties of that element or compound. Molecules are made up of atoms that are held together by chemical bonds.
or
A molecule is an electrically neutral group of two or more atoms held together by chemical bonds.
or
A molecule is two or more atoms bonded together to form a single chemical entity. Each atom carries a certain number of electrons that orbit around the nucleus.
Some examples of molecules include:
- Water: H₂O
- Oxygen: O2
- Ozone: 03
Types of Molecules
A. Diatomic Molecules – A diatomic atom is composed of only two atoms, of the same or different chemical elements. Examples of diatomic molecules are O2 and CO.
B. Heteronuclear Diatomic Molecules – A heteronuclear diatomic molecule consists of two of atoms of the same element combined. There are seven diatomice Elements: Hydrogen (H2), Nitrogen (N₂), Oxygen (O2), Fluorine (F2), Chlorine (C12), Iodine (12) and Bromine (Br2). These seven elements are so reactive that they can be found very often bonded with another atom of the same type.
C. Homonuclear diatomic molecules –A homonuclear diatomic molecule consists of two atoms of different elements chemically combined. Examples of homonuclear diatomic molécules are: carbon monoxide, hydrocholoric acid (HCI), and Hydrogen Flouride (HF)
Types of Biological Molecules
1. Proteins
2. Lipid
3. Carbohydrate
4. Nucleic acid
Molecules Represent
Molecules can be represented with a chemical formula which shows the types of atoms in the molecule, and, uses subscripts, it show how many of each type of atom is present.
A chemical formula is a way of expressing information about the proportions of atoms that constitute a particular chemical compound or molecule, using a single line of chemical element symbols, numbers, and sometimes also other symbols,
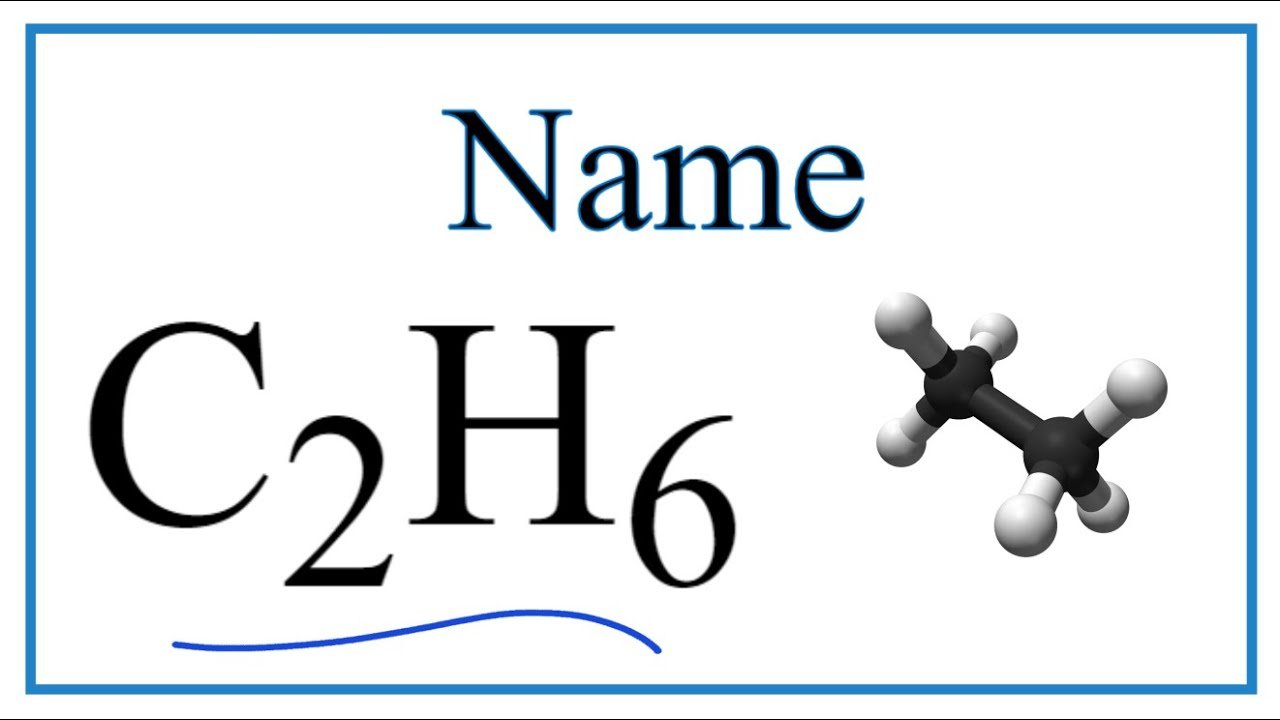
Such as parentheses, dashes, brackets, commas and plus (+) and minus (-) signs. Chemical formulas are most often expressed as –
Molecular formulas which indicate the simple numbers of each type of atom in a molecule of a molecular substance.
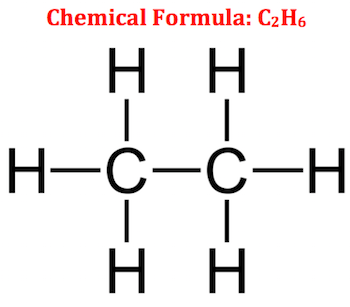
The structural formula of a chemical compound is a graphic representation of the molecular structure, showing how the atoms are arranged.
Difference between Molecule and Compound
| SI. No | Differentiating Property | Molecule | Compound |
| 1 | Definition | A molecule is a group of two or more atoms held together by chemical bonds | A compound is a substance which is form by two or more different types of elemen which are united chemically in a fixed proportion. |
| 2 | Relatedness | All molecules are not compounds. | All compounds are molecules. |
| 3 | Example | An example of a molecule is ozone. (O3) | An example of a compound is table salt (sodium chloride). |
| 4 | Structure | Molecules are simply a group of atoms which are bonded by a strong force. | All compounds are actual matter in their complete shape. |
| 5 | Visibility | Molecule cannot be seen with the naked eye as they are at atomic level. | A compound can be easily seen with the naked eye |
| 6 | Stability | A molecule can be very unstable. | A compound is very stable as it has a phys form. |
